Abstract
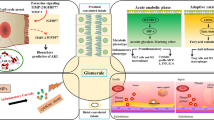
Sepsis is a serious medical condition that can lead to multi-organ failure and shock, and it is associated with increased mortality. Acute kidney injury (AKI) is a frequent complication of sepsis in critically ill patients, and often requires renal replacement therapy. The pathophysiology of AKI in sepsis has not yet been fully defined. In the past, classic theories were mainly focused on systemic hemodynamic derangements, underscoring the key role of whole kidney hypoperfusion due to reduced renal blood flow. However, a growing body of experimental and clinical evidence now shows that, at least in the early phase of sepsis-associated AKI, renal blood flow is normal, or even increased. This could suggest a dissociation between renal blood flow and kidney function. In addition, the scant data available from kidney biopsies in human studies do not support diffuse acute tubular necrosis as the predominant lesion. Instead, increasing importance is now attributed to kidney damage resulting from a complex interaction between immunologic mechanisms, inflammatory cascade activation, and deranged coagulation pathways, leading to microvascular dysfunction, endothelial damage, leukocyte/platelet activation with the formation of micro-thrombi, epithelial tubular cell injury and dysfunction. Moreover, the same processes, through maladaptive responses leading to fibrosis acting from the very beginning, may set the stage for progression to chronic kidney disease in survivors from sepsis-associated AKI episodes. The aim of this narrative review is to summarize and discuss the latest evidence on the pathophysiological mechanisms involved in septic AKI, based on the most recent data from the literature.



Similar content being viewed by others
References
Singer M, Deutschman CS et al (2016) The third international consensus definitions for sepsis and septic shock (Sepsis-3). JAMA 23 315(8):801–810
Rhodes A, Evans LE et al (2017) Surviving sepsis campaign: international guidelines for management of sepsis and septic shock: 2016. Crit Care Med 45(3):486–552
Mehta RL, Kellum JA, Shah SV et al (2007) Acute kidney injury network: report of an initiative to improve outcomes in acute kidney injury. Crit Care 11:31
Bouchard J, Acharya A et al (2015) A Prospective international multicenter study of AKI in the intensive care unit. Clin J Am Soc Nephrol 7;10(8):1324–1331
Lameire NH, Bagga A, Cruz D et al (2013) Acute kidney injury: an increasing global concern. Lancet 13:170–179
Wald R, McArthur E et al (2015) Changing incidence and outcomes following dialysis-requiring acute kidney injury among critically ill adults: a population-based cohort study. Am J Kidney Dis 65(6):870–877
Bellomo R, Kellum JA, Ronco C et al (2017) Acute kidney injury in sepsis. Intensive Care Med 43(6):816–828
Dellepiane S, Marengo M, Cantaluppi V (2016) Detrimental cross-talk between sepsis and acute kidney injury: new pathogenic mechanisms, early biomarkers and targeted therapies. Crit Care 20:61
Gómez H, Kellum JA, Ronco C (2017) Metabolic reprogramming and tolerance during sepsis-induced AKI. Nat Rev Nephrol 13(3):143–151
Langenberg C, Bagshaw SM, May CN et al (2008) The histopathology of septic acute kidney injury: a systematic review. Crit Care 12:R38
Takasu O, Gaut JP, Watanabe E et al (2013) Mechanisms of cardiac and renal dysfunction in patients dying of sepsis. Am J Respir Crit Care Med 187:509–517
Castellano G, Stasi A, Intini A et al. (2014) Endothelial dysfunction and renal fibrosis in endotoxemia-induced oliguric kidney injury: possible role of LPS-binding protein. Crit Care. 18:520–538
Lerolle N, Nochy D, Guérot E et al (2010) Histopathology of septic shock induced acute kidney injury: apoptosis and leukocytic infiltration. Intensive Care Med 36:471–478
Kosaka J, Lankadeva YR, May CN et al (2016) Histopathology of septic acute kidney injury: a systematic review of experimental data. Crit Care Med 44(9):e897-903
Angus DC, van der Poll T (2013) Severe sepsis and septic shock. N Engl J Med 369:2063–2075
Antonucci E, Fiaccadori E et al (2014) Myocardial depression in sepsis: from pathogenesis to clinical manifestations and treatment. J Crit Care 29(4):500–511
Langenberg C, Bellomo R, May C et al (2005) Renal blood flow in sepsis. Crit Care 9:363–374
Chua HR, Glassford N, Bellomo R (2012) Acute kidney injury after cardiac arrest. Resuscitation 83:721–727
Murugan R, Karajala-Subramanyam V, Lee M et al (2010) Acute kidney injury in non-severe pneumonia is associated with an increased immune response and lower survival. Kidney Int 77:527–535
Bradley VE, Shier MR, Lucas CE et al (1976) Renal hemodynamic response to furosemide in septic and injured patients. Surgery 79:549–554
Prowle JR, Molan MP, Hornsey E et al (2012) Measurement of renal blood flow by phase-contrast magnetic resonance imaging during septic acute kidney injury: a pilot investigation. Crit Care Med 40(6):1768–1776
Badr KF, Kelley VE, Rennke HG et al (1986) Roles for thromboxane A2 and leukotrienes in endotoxin-induced acute renal failure. Kidney Int 30(4):474–480
Ravikant T, Lucas CE (1977) Renal blood flow distribution in septic hyperdynamic pigs. J Surg Res 22:294–298
Langenberg C, Wan L, Egi M et al (2007) Renal blood flow and function during recovery from experimental septic acute kidney injury. Intensive Care Med 33:1614–1618
Maiden MJ, Otto S et al (2016) Structure and function of the kidney in septic shock. A prospective controlled experimental study. Am J Respir Crit Care Med 194(6):692–700
De Backer D, Donadello K, Taccone FS et al (2011) Microcirculatory alterations: potential mechanisms and implications for therapy. Ann Intensive Care 19:27–35
Holthoff JH, Wang Z, Seely KA et al. (2012) Resveratrol improves renal microcirculation, protects the tubular epithelium, and prolongs survival in a mouse model of sepsis-induced acute kidney injury. Kidney Int. 81:370–378
Dyson A, Bezemer R, Legrand M et al. (2011) Microvascular and interstitial oxygen tension in the renal cortex and medulla studied in a 4-h rat model of LPS-induced endotoxemia. Shock 36:83–89
Legrand M, Bezemer R, Kandil A et al (2011) The role of renal hypoperfusion in development of renal microcirculatory dysfunction in endotoxemic rats. Intensive Care Med 37:1534–1542
Losser MR, Forget AP, Payen D (2006) Nitric oxide involvement in the hemodynamic response to fluid resuscitation in endotoxic shock in rats. Crit Care Med 34:2426–2431
Heemskerk S, Masereeuw R, Russel FG et al (2009) Selective iNOS inhibition for the treatment of sepsis-induced acute kidney injury. Nat Rev Nephrol 5:629–640
Langenberg C, Gobe G, Hood S et al (2014) Renal histopathology during experimental septic acute kidney injury and recovery. Crit Care Med 42:58–67
Legrand M, Dupuis C, Simon C et al (2013) Association between systemic hemodynamics and septic acute kidney injury in critically ill patients: a retrospective observational study. Crit Care 17(6):R278
Gustot T (2011) Multiple organ failure in sepsis: prognosis and role of systemic inflammatory response. Curr Opin Crit Care 17:153–159
Jo SK, Cha DR, Cho WY et al (2002) Inflammatory cytokines and lipopolysaccharide induce Fas-mediated apoptosis in renal tubular cells. Nephron 91(3):406–415
Lee SY, Lee YS, Choi HM et al (2012) Distinct pathophysiologic mechanisms of septic acute kidney injury: role of immune suppression and renal tubular cell apoptosis in murine model of septic acute kidney injury. Crit Care Med 40:2997–2906
Mariano F, Cantaluppi V et al (2008) Circulating plasma factors induce tubular and glomerular alterations in septic burns patients. Crit Care 12(2):R42
Gomez H, Ince C, De Backer D et al (2014) A unified theory of sepsis-induced acute kidney injury: inflammation, microcirculatory dysfunction, bioenergetics, and the tubular cell adaptation to. injuryShock 41:3–11
Castoldi A, Braga TT, Correa-Costa M et al (2012) TLR2, TLR4 and the MYD88 signaling pathway are crucial for neutrophil migration in acute kidney injury induced by sepsis. PLoS One 7(5)
Knotek M, Rogachev B, Wang W et al (2001) Endotoxemic renal failure in mice: role of tumor necrosis factor independent of inducible nitric oxide synthase. Kidney Int 59:2243–2249
Cunningham PN, Wang Y, Guo R et al (2004) Role of Toll-like receptor 4 in endotoxin-induced acute renal failure. J Immunol 172:2629–2635
Benes J, Chvojka J, Sykora R et al (2011) Searching for mechanisms that matter in early septic acute kidney injury: an experimental study. Crit Care 15:256–263
Rodriguez-Wilhelmi P, Montes R, Matsukawa A et al (2003) Tumor necrosis factor-alpha inhibition reduces CXCL-8 levels but fails to prevent fibrin generation and does not improve outcome in a rabbit model of endotoxic shock. J Lab Clin Med 141:257
Cantaluppi V, Gatti S et al (2012) Microvesicles derived from endothelial progenitor cells protect the kidney from ischemia-reperfusion injury by microRNA-dependent reprogramming of resident renal cells. Kidney Int 82(4):412–427
Zafrani L, Gerotziafas G, Byrnes C et al (2012) Calpastatin controls polymicrobial sepsis by limiting procoagulant microparticle release. Am J Respir Crit Care Med 185:744–755
Meziani F, Delabranche X, Asfar P et al. (2010) Bench-to-bedside review: circulating microparticles–a new player in sepsis?. Crit Care 14:236–242
Mortaza S, Martinez MC, Baron-Menguy C et al (2009) Detrimental hemodynamic and inflammatory effects of microparticles originating from septic rats. Crit Care Med 37:2045–2050
Zafrani L, Ince C, Yuen PS (2013) Microparticles during sepsis: target, canary or cure? Intensive Care Med 39(10):1854–1856
Levi M, De Jonge E, Van der Poll T (2001) Rationale for restoration of physiological anticoagulant pathways in patients with sepsis and disseminated intravascular coagulation. Crit Care Med 29(7 Suppl):S90-4
Engelmann B, Massberg S (2013) Thrombosis as an intravascular effector of innate immunity. Nat Rev Immunol 13(1):34–45
Levi M, Van der Poll T (2005) Two-way interactions between inflammation and coagulation. Trends Cardiovasc Med 15(7):254–259
Suárez-Álvarez B, Liapis H, Anders HJ (2016) Links between coagulation, inflammation, regeneration, and fibrosis in kidney pathology. Lab Invest 96(4):378–390
Forni LG, Darmon M et al (2017) Renal recovery after acute kidney injury. Intensive Care Med 43(6):855–866
Sawhney S, Marks A et al (2017) Post-discharge kidney function is associated with subsequent ten-year renal progression risk among survivors of acute kidney injury. Kidney Int 92(2):440–452
Rabb H, Griffin MD et al (2016) Inflammation in AKI: current understanding, key questions, and knowledge gaps. J Am Soc Nephrol 27(2):371–379
Kaballo MA, Elsayed ME, Stack AG (2017) Linking acute kidney injury to chronic kidney disease: the missing links. J Nephrol 30(4):461–475
Ferenbach DA, Bonventre JV (2015) Mechanisms of maladaptive repair after AKI leading to accelerated kidney ageing and CKD. Nat Rev Nephrol. 11(5):264–276
Deng W, Cho S et al (2014) Membrane-enabled dimerization of the intrinsically disordered cytoplasmic domain of ADAM10. Proc Natl Acad Sci USA 111(45):15987–15992
Kulkarni OP, Hartter I et al. (2014) Toll-like receptor 4-induced IL-22 accelerates kidney regeneration. J Am Soc Nephrol 25(5):978–89
Collino F, Bruno S et al (2015) AKI recovery induced by mesenchymal stromal cell-derived extracellular vesicles carrying microRNAs. J Am Soc Nephrol 26(10):2349–2360
Basile DP, Bonventre JV et al (2016) Progression after AKI: understanding maladaptive repair processes to predict and identify therapeutic treatments. J Am Soc Nephrol 27(3):687–697
Zuk A, Bonventre JV (2016) Acute kidney injury. Annu Rev Med 67:293–307
Cianciolo Cosentino C, Skrypnyk NI et al. (2013) Histone deacetylase inhibitor enhances recovery after AKI. J Am Soc Nephrol. 24(6):943–953
Wynn TA (2010) Fibrosis under arrest. Nat Med 16(5):523–525
Basile DP (2007) The endothelial cell in ischemic acute kidney injury: implications for acute and chronic function. Kidney Int 72(2):151–156
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Human or animals participant statements
This article does not contain any studies with animals or human participants performed by any of the authors.
Funding
This paper was partially funded by the Progetto Giovani Ricercatori Bando 2011–2012, funding number Prot. 2010J4458Z.
Rights and permissions
About this article
Cite this article
Fani, F., Regolisti, G., Delsante, M. et al. Recent advances in the pathogenetic mechanisms of sepsis-associated acute kidney injury. J Nephrol 31, 351–359 (2018). https://doi.org/10.1007/s40620-017-0452-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40620-017-0452-4